 
First law of thermodynamics calculator code#Compute properties of ideal gases, examine the evolution of systems under thermodynamic processes and calculate thermodynamic properties of chemical substances. method for calculating HEX with a thermodynamic code has been described. The relationship between the energy change of a system and that of its surroundings is given by. Use WolframAlpha to explore the consequences of the laws of thermodynamics. On solving W = 2 k x 2 + F a t m = 2 k x 2 2 + F a t m x 2 . Thermodynamics studies heat and temperature and how they interact with the energy flow of systems. This puts a constraint on the heat engine. W = x 1 = 0 ∫ x 2 F t o t a l d x = x 1 = 0 ∫ x 2 ( F s p r i n g + F a t m ) d x. The first law of thermodynamics with Q0 shows that all the change in internal energy is in the form of work done. The spring force varies linearly with position.įor a quasi-equilibrium process, boundary or P-V work is defined by : The process occurs slowly enough that it is a quasi-equilibrium process.ģ. There is no friction between the piston and the cylinder wall.Ĥ. 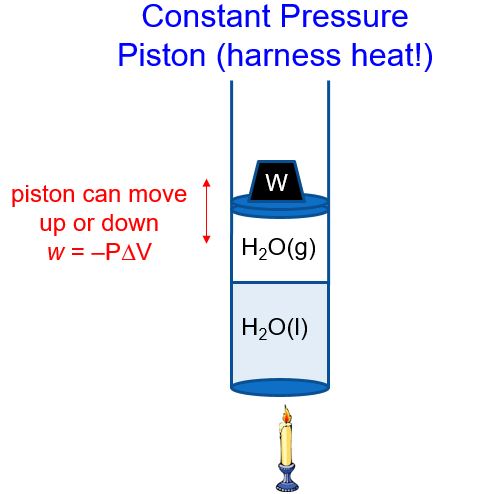
The air in the cylinder is a closed system.Ģ. Here, U is the change in internal energy, U, of the system. In equation form, the first law of thermodynamics is 12.6 U Q W. P a t m = 7 5 k P a k = 1 k N / m W = ? k Jġ. The first law of thermodynamics states that the change in internal energy of a closed system equals the net heat transfer into the system minus the net work done by the system. Given P 2 = 4 0 0 k P a D P i s t o n = 0. First law of thermodynamics calculator plus#This relationship is linear because the pressure within the cylinder is atmospheric pressure plus the spring force divided by the cross-sectional area of the piston. It shows the relationship between internal energy, heat, a. Key Note : The key to solving this problem is to determine the slope and intercept for the linear relationship between the force exerted by the spring on the piston and the pressure within the gas. This chemistry video tutorial provides a basic introduction into the first law of thermodynamics. Determine the work done by the gas on the piston. Isobaric Process and the First Law The classical form of the first law of thermodynamics is the following equation: dU dQ dW In this equation, dW is equal to dW pdV and is known as the boundary work. 
Energy is added to the gas inside the cylinder until the pressure in the cylinder is 4 0 0 k P a. An unstretched spring is attached to a horizontal, frictionless piston. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
